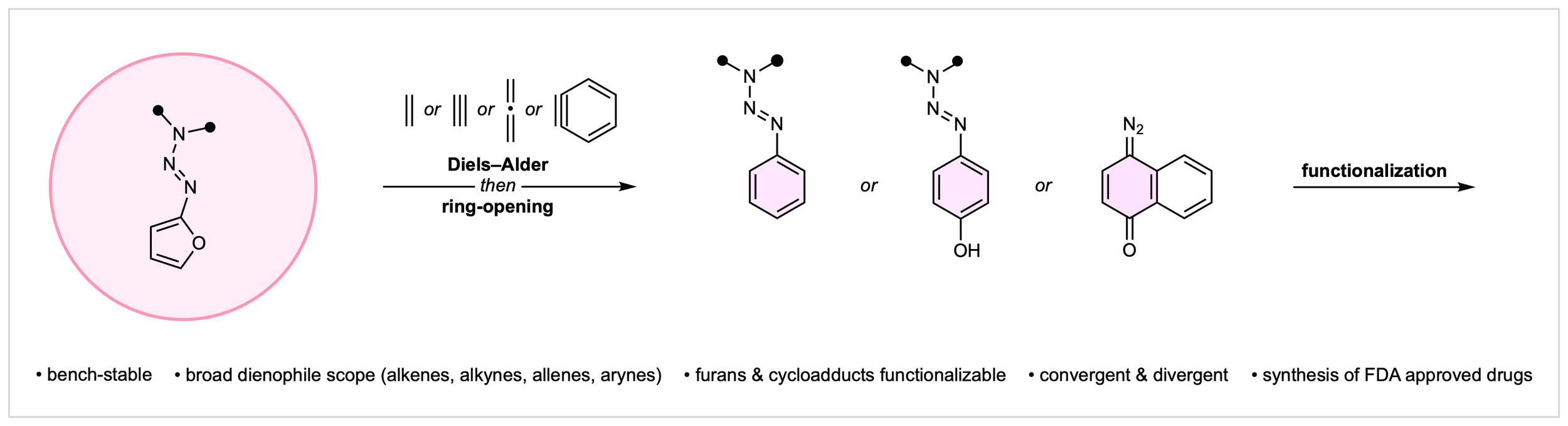
26. Triazenyl Furans as Diels–Alder Dienes
J. E. Budwitz,§ S. A. Hollers,§ A. R. Wester, C. G. Newton*
submitted
§ Equal Contribution
• Preprint available on ChemRxiv, DOI: 10.26434/chemrxiv.15001641/v1
• Highlighted in: Synthesis Spotlight, Monday 6th April – Sunday 12th April 2026, Volume 3, Issue 14
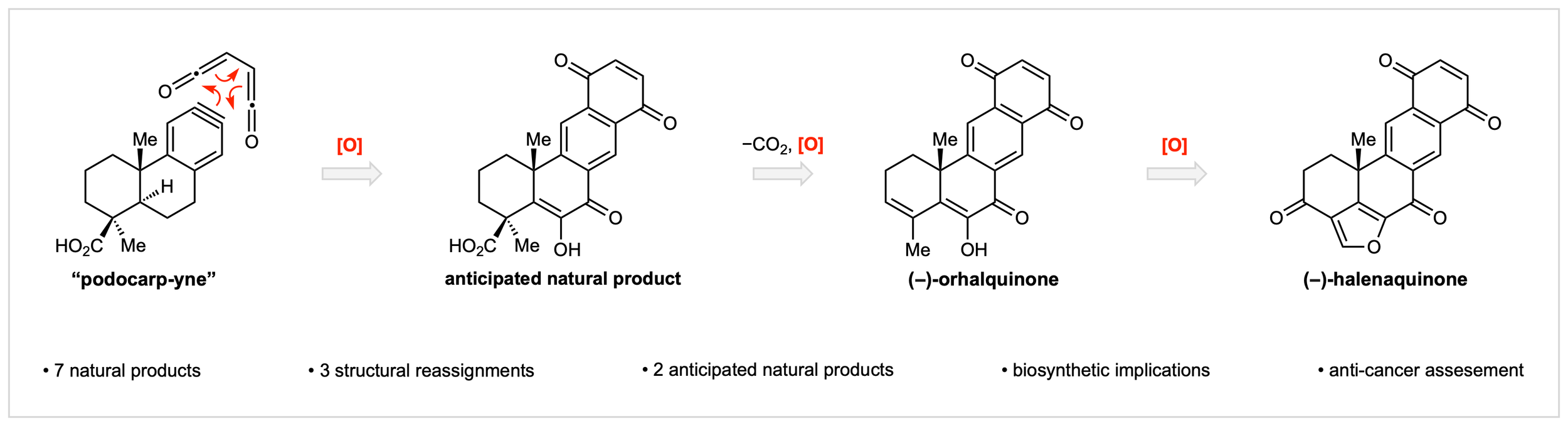
25. Biomimetic Synthesis of Seven Halenaquinone Meroterpenoids
J. D. Hart,§ J. K. Lawrence,§ P. N. Franqui, D. R. Perrott, B. Yi, Y. Xi, C. J. Sumby, C. G. Newton,* J. H. George*
submitted
§ Equal Contribution
• Preprint available on ChemRxiv, DOI: 10.26434/chemrxiv.15001574/v1
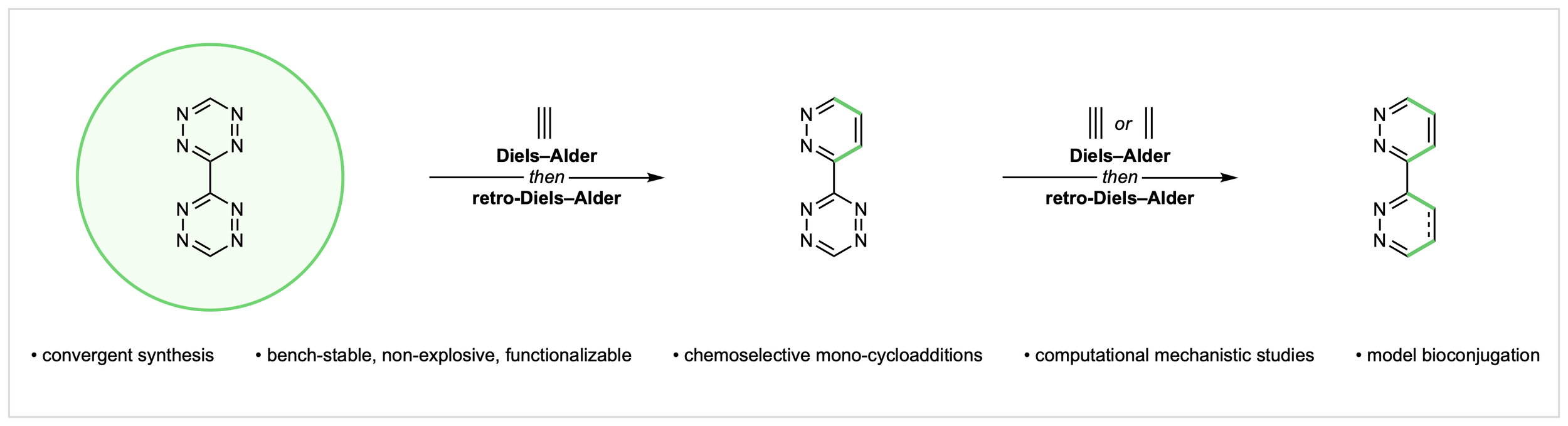
24. Biaryl Tetrazines as Chemoselective Double-Click Reagents
M. J. Liss,§ G. Goyal,§ A. V. Conner, S. E. Wheeler,* C. G. Newton*
submitted
§ Equal Contribution
• Preprint available on ChemRxiv, DOI: 10.26434/chemrxiv.15001230/v1
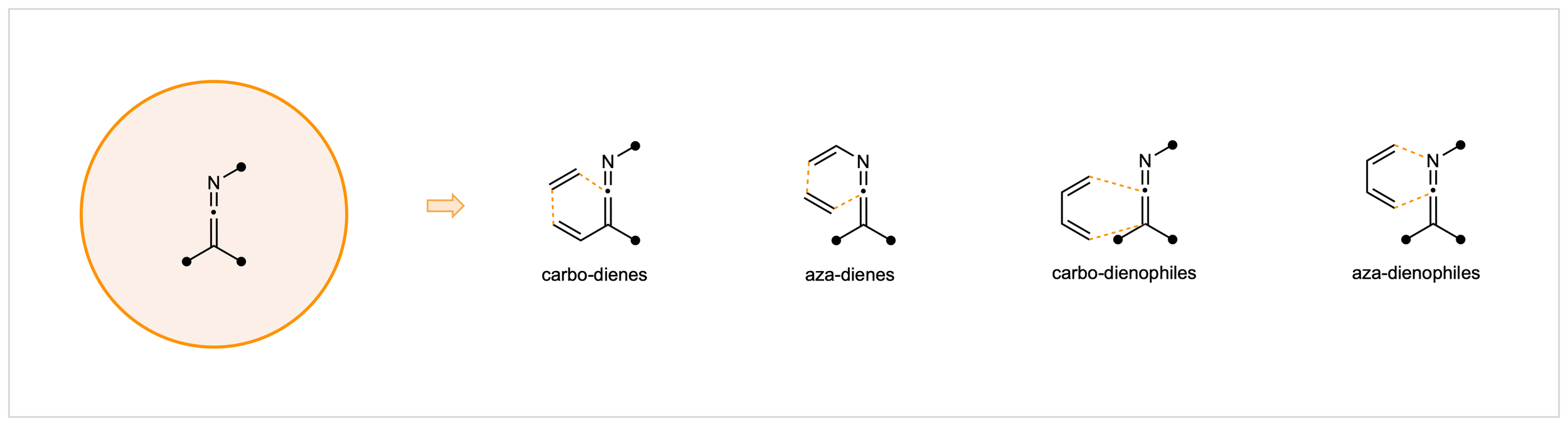
23. Diels–Alder Chemistry of Ketenimines: Toward Highly Reactive Aza-Dienophiles
M. J. Liss, C. G. Newton*
Synlett, 2026, in press. DOI: 10.1055/a-2850-9697
• Invited “Synpacts” Article
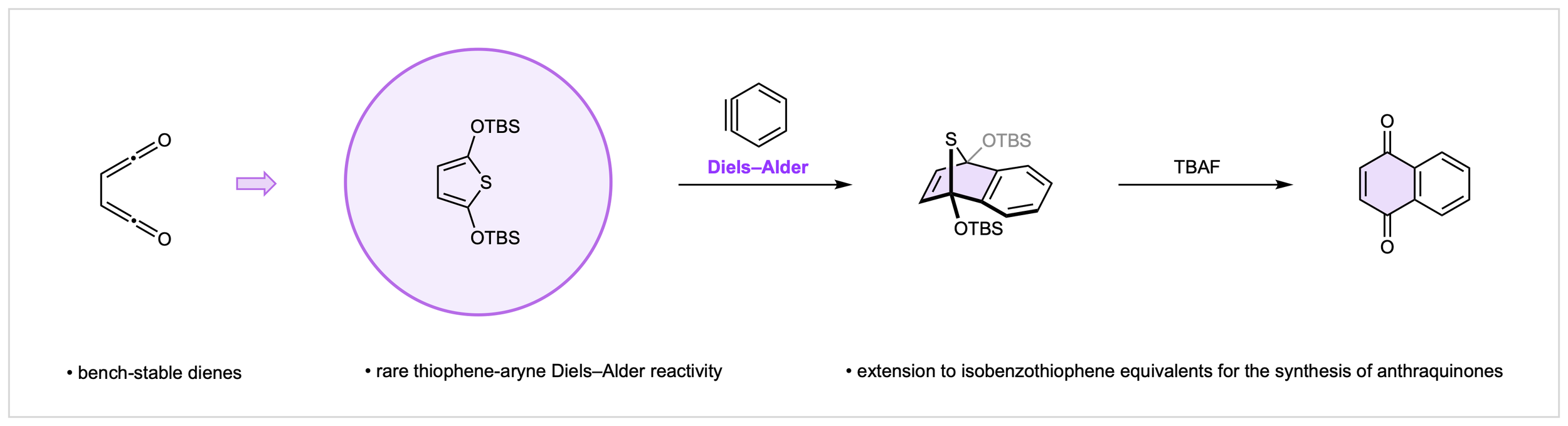
22. Stable Vicinal Bisketene Equivalents for Aryne Diels–Alder Reactions
J. E. Budwitz, C. G. Newton*
Synlett, 2026, in press. DOI: 10.1055/a-2779-2027
• Invited Submission as part of the “Alkynes in Organic Synthesis” Special Issue
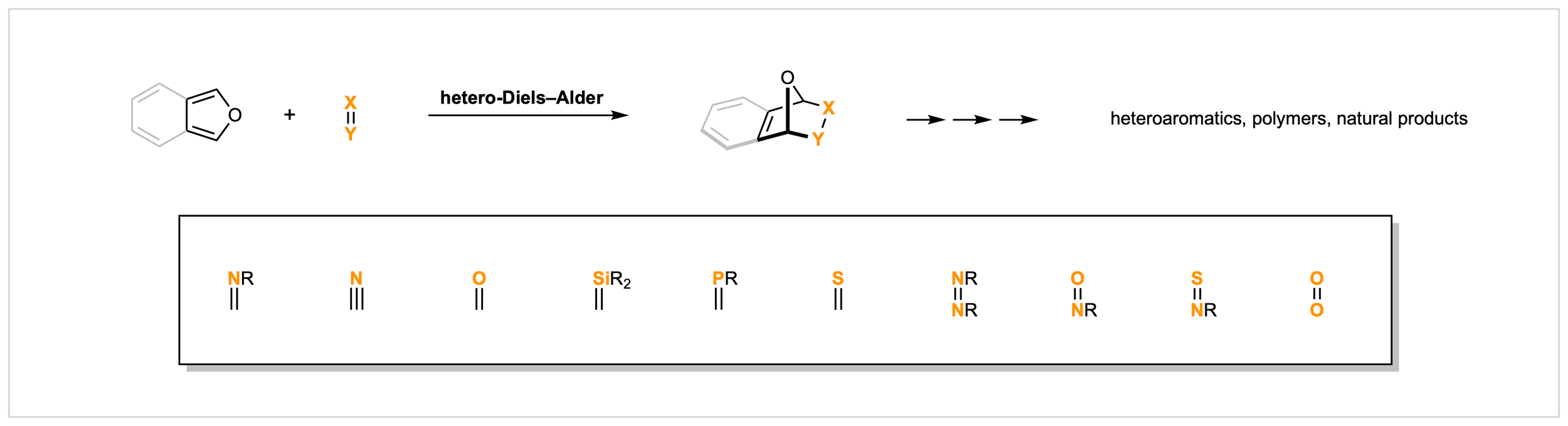
21. Hetero-Diels–Alder Reactions of (Isobenzo)Furans
C. J. DeAngelis, C. G. Newton*
Org. Biomol. Chem. 2025, 23, 9781
• Invited Submission as part of the "2025 New Talent Issue”
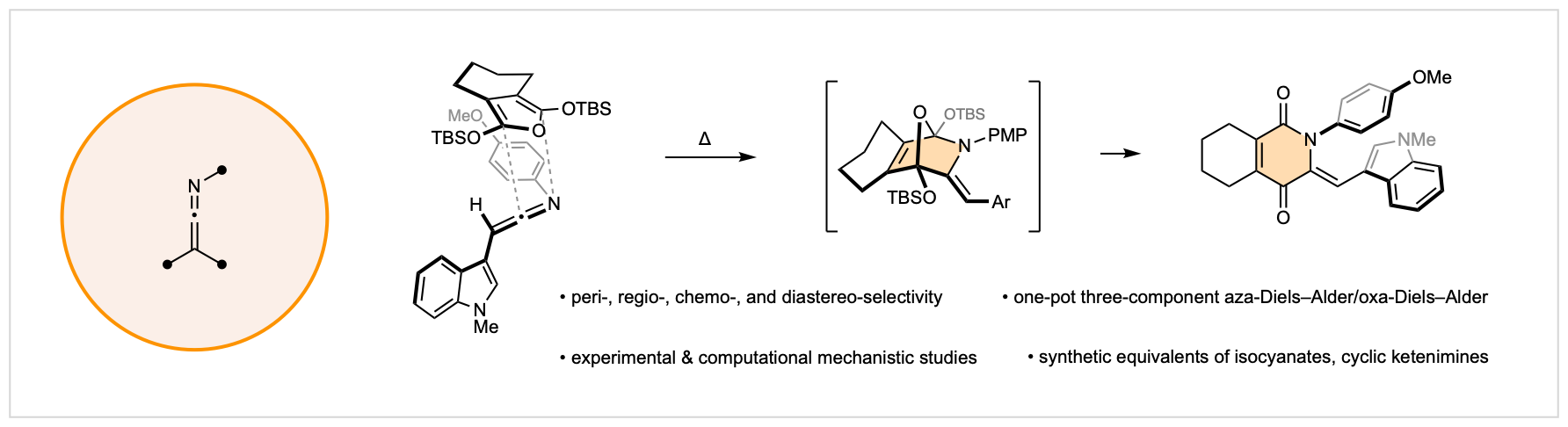
20. Ketenimines as Aza-Dienophiles
C. J. DeAngelis,§ G. Goyal,§ M. J. Liss,§ J. E. Budwitz, M. S. Herlihy, A. V. Conner, S. E. Wheeler, P. Ma, M. Li, K. N. Houk,* C. G. Newton*
J. Am. Chem. Soc. 2025, 147, 6087
§ Equal Contribution
• Preprint available on ChemRxiv, DOI: 10.26434/chemrxiv-2024-7rvht

19. Synthesis of a 2,5-Bis(tert-butyldimethylsilyloxy)furan and its Reaction with Benzyne
J. E. Budwitz, C. G. Newton*
Org. Synth. 2023, 100, 159
• Invited Submission
18. Early Career Author Profile
C. G. Newton*
Helv. Chim. Acta, 2023, 106, e202300007

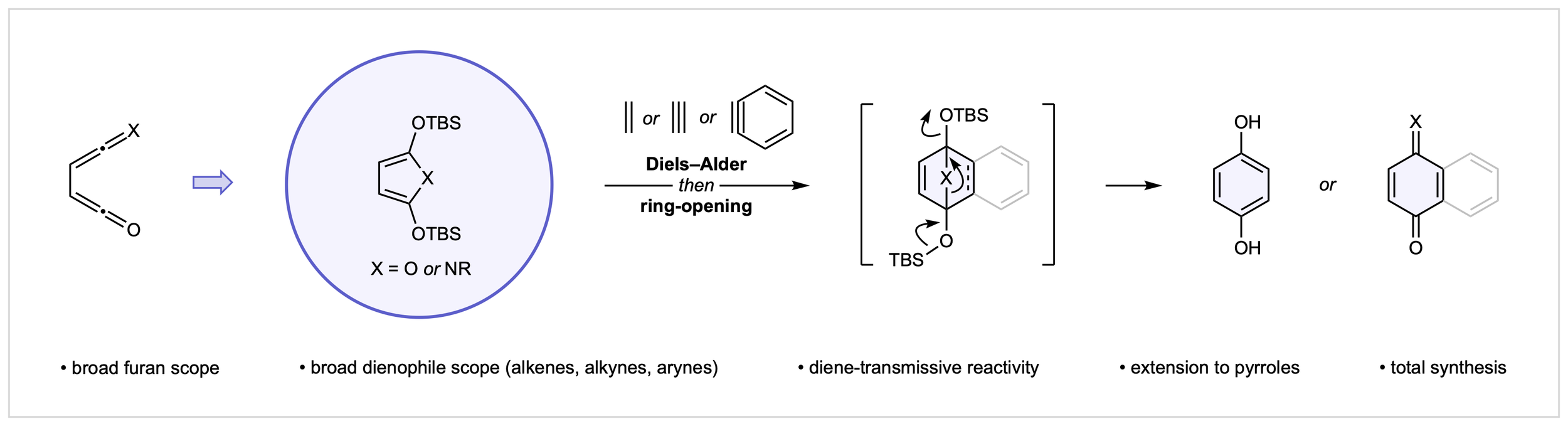
17. Ring Forming Approaches to para-Quinones: Toward a General Diels–Alder Disconnection
J. E. Budwitz, C. G. Newton*
Synlett, 2022, 33, 1473
• Invited “Synpacts” Article
16. Bisketene Equivalents as Diels–Alder Dienes
I. Dissanayake,§ J. D. Hart,§ E. C. Becroft, C. J. Sumby, C. G. Newton*
J. Am. Chem. Soc. 2020, 142, 13328
§ Equal Contribution
• Preprint available on ChemRxiv, DOI: 10.26434/chemrxiv.12463949
• Highlighted in: ChemViews Mag.2020, DOI: 10.1002/chemv.202000072
• Highlighted in: Chemistry in Australia, September–November 2020, 12
15. Network Analysis of Substituted Bullvalenes
O. Yahiaoui, L. F. Pašteka,* C. J. Blake, C. G. Newton, T. Fallon*
Org. Lett. 2019, 21, 9574
14. Visible Light Photoredox Catalysis Enables the Biomimetic Synthesis of Nyingchinoids A, B and D, and Rasumatranin D
J. D. Hart, L. Burchill, A. J. Day, C. G. Newton, C. J. Sumby, D. M. Huang, J. H. George*
Angew. Chem. Int. Ed. 2019, 58, 2791
Pre- and Post-Doctoral
13. A Broad-Spectrum Synthesis of Tetravinylethylenes
K. L. Horvath, C. G. Newton, K. A. Roper, J. S. Ward, M. S. Sherburn
Chem. Eur. J. 2019, 25, 4072
12. Chiral Cp Ligands for Rhodium(III)-Catalyzed Asymmetric Carbon−Hydrogen Bond Functionalization
C. G. Newton, N. Cramer
in Rhodium Catalysis in Organic Synthesis: Methods and Reactions, Wiley-VCH, Weinheim, 2019
11. Axially Chiral Dibenzazepinones by a Palladium(0)‐Catalyzed Atropo‐enantioselective C−H Arylation
C. G. Newton, E. Braconi,§ J. Kuziola,§ M. D. Wodrich, N. Cramer*
Angew. Chem. Int. Ed. 2018, 57, 11040
§ Equal Contribution
10. One‐Step Multigram‐Scale Biomimetic Synthesis of Psiguadial B
C. G. Newton, D. N. Tran, M. D. Wodrich, N. Cramer*
Angew. Chem. Int. Ed. 2017, 56, 13776
9. Catalytic Enantioselective Transformations Involving C−H Bond Cleavage by Transition-Metal Complexes
C. G. Newton,§ S.-G. Wang,§ C. C. Oliveira, N. Cramer*
Chem. Rev. 2017, 117, 8908
§ Equal Contribution
8. Masked Ketenes as Dienophiles in the Diels–Alder Reaction
E. G. Mackay,* C. G. Newton*
Aus. J. Chem. 2016, 69, 1365
7. Cross-Conjugation in Synthesis
C. G. Newton, M. S. Sherburn*
in Cross Conjugation: Modern Dendralene, Radialene and Fulvene Chemistry, Wiley-VCH, Weinheim, 2016
6. Asymmetric Catalysis Powered by Chiral Cyclopentadienyl Ligands
C. G. Newton, D. Kossler, N. Cramer*
J. Am. Chem. Soc. 2016, 138, 3935
5. [5]Radialene
E. G. Mackay, C. G. Newton, H. Toombs-Ruane, E. J. Lindeboom, T. Fallon, A. C. Willis, M. N. Paddon-Row,* M. S. Sherburn*
J. Am. Chem. Soc. 2015, 137, 14653
4. Total Synthesis of the Pseudopterosin Aglycones
C. G. Newton, M. S. Sherburn*
Nat. Prod. Rep. 2015, 32, 865
3. Pseudopterosin Synthesis From a Chiral Cross-Conjugated Hydrocarbon Through a Series of Cycloadditions
C. G. Newton, S. L. Drew, A. L. Lawrence, A. C. Willis, M. N. Paddon-Row,* M. S. Sherburn*
Nat. Chem. 2015, 7, 82
2. 1,1-Divinylallene
K. M. Cergol, C. G. Newton, A. L. Lawrence, A. C. Willis, M. N. Paddon-Row,* M. S. Sherburn*
Angew. Chem. Int. Ed. 2011, 50, 10425
1. The Anti-Cancer, Anti-Inflammatory and Tuberculostatic Activities of a Series of 6,7-Substituted-5,8-Quinolinequinones
B. J. Mulchin, C. G. Newton, J. W. Baty, C. H. Grasso, W. John Martin, M. C. Walton, E. M. Dangerfield, C. H. Plunkett, M. V. Berridge, J. L. Harper, M. S. M. Timmer,* B. L. Stocker*
Bioorg. Med. Chem. 2010, 18, 3238